Service hotline
86-18561281819Application Cases
 Current Location:
Current Location: Quick navigation
Diamond is a mineral composed of carbon element. It is a Allotropy of graphite, with chemical formula C, and is also the origin of common diamonds. Diamond is the hardest substance naturally present in nature. Graphite can form artificial diamonds under high temperature and pressure. Diamond has a wide range of uses, such as handicrafts, cutting tools in industry, and is also a precious gemstone.
Diamond is a type of elemental crystal composed of carbon elements formed under high-pressure and high-temperature conditions deep in the Earth. Diamond is a colorless Regular octahedron crystal. Its component is pure carbon, which is linked by carbon atoms with four valence bonds. It is the hardest substance known to exist naturally. Because the C-C bond in diamond is very strong, all Valence electron participate in the formation of Covalent bond, without free electrons, so the hardness of diamond is very high, the melting point is 6900 degrees Fahrenheit, the ignition point of diamond in pure oxygen is 720~800 ℃, and in air is 850~1000 ℃, and it is non-conductive.
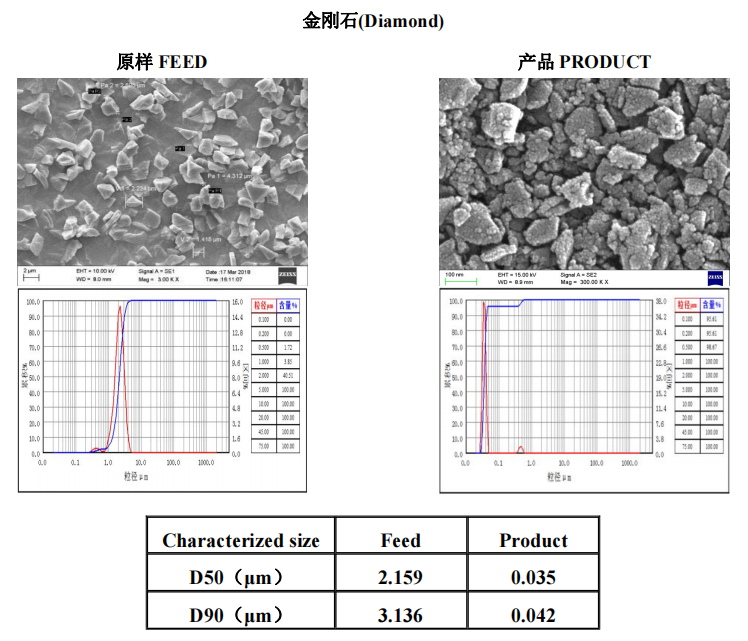
Recommended equipment: SP series bead mill without shaft seal and screen